One-time Revolution - How A Single-use Biopsy Needle Is Shaping Infection Control And Healthcare Economics
Apr 24, 2026
One-time Revolution - How a Single-use Biopsy Needle Is Shaping Infection Control and Healthcare Economics
Key words: One-time biopsy needle system + Eliminate cross-infection and simplify operation procedures
At the intersection of medical safety and health economics, disposable biopsy needles are shifting from an "optional" choice to a "mandatory" one. The driving force behind this transformation is not only the rigid demand for infection control, but also the deep restructuring of the standardization of biopsy procedures, cost transparency, and medical efficiency. From the traditional "reusable disinfected needles" to the modern "one-time use all-inclusive system", the single-use mode of biopsy needles is redefining the quality control standards and economic models of puncture operations.
The data on infection control is alarming. Although high-level disinfection can kill the vast majority of pathogens, the complex internal structure of biopsy needles (such as the tiny gaps between the needle core and the cannula, and the sharp corners of the cutting grooves) provides a place for biofilm formation. The US FDA adverse event database shows that from 2019 to 2023, a total of 127 suspected infections related to reused biopsy needles were reported, of which 42 were infections caused by resistant bacteria. The most famous case was that a medical center, due to insufficient cleaning of prostate biopsy needles, caused the spread of extended-spectrum β-lactamase (ESBL) Escherichia coli among 12 patients. Theoretical calculations show that even if the disinfection effect reaches 99.999% (5-log reduction), when the microbial load of a single biopsy is 10⁶ CFU, the residual microorganisms on reused needles still have 10 CFU, posing a risk to immunocompromised patients. Disposable biopsy needles reduce the risk of cross-infection from a theoretical possibility to zero, which is a safety margin that the reuse system cannot achieve.
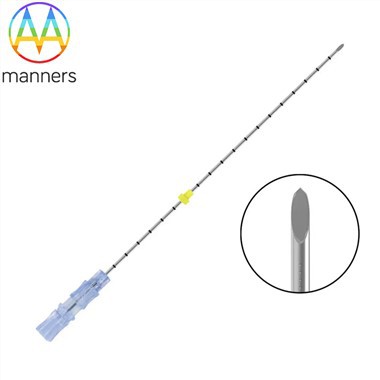
The standardization revolution of the operation process enhances medical quality. The traditional operation of reusing biopsy needles requires seven steps: cleaning, soaking, ultrasonic treatment, rinsing, drying, packaging, and sterilization. Any mistake at any step can affect the sterilization effect. The "open and use immediately" feature of disposable biopsy needles eliminates these seven variables. A more profound change lies in the development of the fully integrated biopsy system: an aseptic packaging contains the biopsy needle, positioning guide wire, tissue preservation solution, sample bottle, pathology application form, and even the patient identification code, achieving a closed loop of "one patient, one biopsy, one package". Clinical studies show that using the fully integrated system, biopsy-related errors (such as sample identification errors, improper fixation, etc.) have decreased from 3.2 times per thousand cases to 0.4 times, and the sample return rate has dropped from 2.1% to 0.3%. In terms of time efficiency, the average time from deciding on the biopsy to completing the puncture has been shortened from 45 minutes (including preparation time) with the reusable system to 18 minutes.
The redefinition of cost structure goes beyond simple comparison. On the surface, the one-time purchase cost of a disposable biopsy needle is higher than the "single-use cost" of a reusable needle (the purchase price divided by the number of uses). However, a full cost analysis reveals a different picture: the true cost of the reuse system includes initial purchase, cleaning consumables, disinfection equipment depreciation, labor time, quality inspection, maintenance, and final disposal. The detailed cost accounting at the Mayo Clinic in the United States shows that the single real cost of a reusable biopsy needle that can be used 20 times is $87.5, with the direct purchase cost accounting for only 28%, cleaning and disinfection accounting for 42%, and management and maintenance accounting for 30%. Meanwhile, a disposable needle of the same performance has a purchase price of $79. Disposable needles also avoid biopsy interruptions caused by needle damage (with an occurrence rate of approximately 0.8%), and the average loss per interruption (operating room occupancy, doctor time, patient anxiety) amounts to $350. From a hospital financial perspective, disposable needles convert uncertain operating costs into certain purchase costs, which is more conducive to budget management.
Innovation in environmental protection and sustainability has become the focus of the industry. Critics point out that disposable medical devices increase medical waste. The industry's response is multi-faceted. In terms of materials, the new biopsy needles use recyclable polypropylene (label #5) and biobased polymers, with a bio-carbon content of 30%. The weight of a single needle has decreased from the early 18g to 9g. In terms of packaging, recyclable paper-plastic is used instead of the non-recyclable Tyvek bag, reducing the packaging volume by 40%. Closed-loop recycling projects are being promoted in Europe and the United States: used biopsy needles (sterilized under high pressure) are classified and recycled, with the polymer part processed into municipal building materials (such as park benches), and the metal part melted and reused. The "environmental impact index" of leading manufacturers shows that the carbon footprint of their disposable biopsy needles throughout their life cycle is 15% lower than the reusable system, mainly due to the huge energy consumption of the high-temperature and high-pressure sterilization process of the reusable system.
The special value in resource-limited environments is often overlooked. In grassroots hospitals, field hospitals, and disaster medical scenarios, the cleaning and disinfection facilities required for reusable systems are often lacking. Disposable biopsy needles can be used under basic aseptic conditions. The WHO's "Basic Medical Equipment List" has classified disposable biopsy needles as a Class B recommendation (highly recommended under specific conditions). In the African malaria research project, the use of disposable liver biopsy needles enabled grassroots health centers to safely conduct malaria liver-stage research, with sample quality comparable to that of tertiary hospitals. Adaptive design for extreme environments has emerged: biopsy needles that can be stored at -20°C to 50°C, suitable for polar expeditions and tropical regions; small packages with built-in desiccants that can be preserved for 3 years in high-humidity environments.
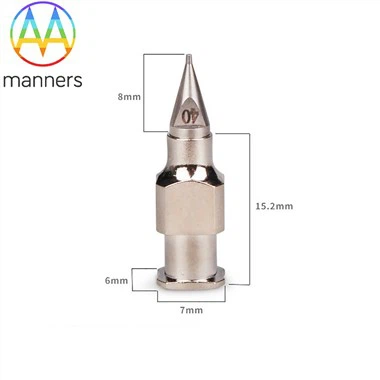
The evolution of regulations and standards is accelerating the transformation. The 2024 new regulations of the US FDA require that all "reusable medical devices that come into contact with human tissues" must provide detailed data on "cleaning verification" to prove that they can meet the disinfection standards even under the most adverse conditions (such as delayed cleaning for 6 hours after a biopsy). This poses a huge challenge for biopsy needle manufacturers - the cleaning verification of a tube with an inner diameter of only 1mm is extremely difficult. The EU MDR regulations require that reusable devices undergo "use-life verification", and biopsy needles must prove that they remain structurally intact and functionally normal after being disinfected 200 times. These regulations essentially drive the process towards one-time use. The industry standard ISO 23908:2024 specifically regulates the performance consistency of disposable biopsy needles, requiring that the cutting force difference of the same batch of needles be <15% and the sample weight difference be <20%.
In the future, disposable biopsy needles will evolve into intelligent disposable systems. The needle handle integrates an RFID chip to record the production batch number, expiration date, and sterilization date; after scanning, it automatically enters the hospital information system. The needle tip pressure sensor provides tactile feedback when puncturing abnormal tissues (such as calcification foci). After use, the color developer inside the needle changes color upon contact with blood, ensuring that it cannot be reused maliciously. By 2027, the global disposable biopsy needle market will reach 6.4 billion US dollars, with the penetration rate increasing from the current 45% to 70%. The ultimate goal of this "disposable revolution" is to provide each patient with an absolutely safe, absolutely standard, and absolutely exclusive biopsy experience without increasing the total medical cost, and to upgrade medical safety from "probability control" to "certainty guarantee".