Analysis Of Customized Service Model For Laparoscopic Trocar Manufacturers
May 04, 2026
Against the backdrop of increasingly fierce competition in the medical device industry, customized services have become an important means for laparoscopic trocar manufacturers to differentiate themselves. From OEM to ODM, and from standardized production to flexible manufacturing, manufacturers are meeting the diverse needs of customers through diversified service models and building their core competitiveness.
The development background and market demand of customized services
The diversification and personalization trends of laparoscopic surgery have driven the demand for customized trocars. Different surgical types (general surgery, gynecology, urology, etc.) require trocars of different specifications; the anatomical differences among patients call for personalized adjustments in the size and shape of trocars; and the innovation of surgical techniques (such as single-port laparoscopy, robot-assisted surgery) demands specially designed trocar instruments.
The enhanced focus of medical institutions on cost control and quality requirements has also promoted the development of customized services. Public hospitals usually purchase through centralized bidding and are sensitive to prices; private hospitals and high-end medical institutions, on the other hand, pay more attention to product performance and service quality. Manufacturers need to offer flexible product solutions and service models to meet the diverse needs of different clients.
The differences in global medical device regulatory systems have increased market complexity. Major countries and regions such as China, the United States, the European Union, and Brazil have different medical device registration requirements and quality standards. Manufacturers need to help customers address these regulatory challenges and provide products and services that meet local standards.
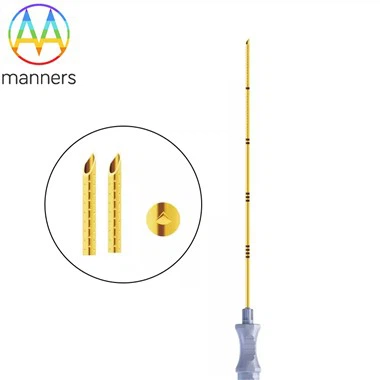
OEM Service Model: Precision Manufacturing According to Drawings
OEM (Original Equipment Manufacturer) is the fundamental model for customized laparoscopic trocar services. Under this model, the client provides detailed product design drawings and technical requirements, and the manufacturer strictly produces according to the client's specifications. Haina Medical has accumulated rich experience in this field and has provided high-precision core parts for international giants such as Johnson & Johnson and Medtronic.
The core advantage of OEM services lies in manufacturing specialization and cost control. Manufacturers focus on the production process, reducing manufacturing costs through large-scale production and process optimization. Cleanrooms of 100,000 grades, automated production lines, and artificial intelligence quality inspection systems ensure that the defect rate of products is less than 0.1%. Stringent quality control management throughout the entire process, from raw material procurement to finished product delivery, has strict inspection standards for each link.
Rapid response to sample order demands is an important capability of OEM services. The development of medical device products often requires multiple iterations, and small-batch trial production and large-scale production need to be seamlessly connected. Manufacturers need to establish flexible production lines that can quickly switch product models to adapt to the product development pace of customers.
ODM service model: One-stop solution from design to manufacturing
The ODM (Original Design Manufacturer) service model offers higher value-added customized services. Manufacturers are not only responsible for production but also participate in the entire process of product design, development and testing. This model is particularly suitable for start-up enterprises and small and medium-sized medical device companies with limited resources.
The comprehensive medical device development and manufacturing platform of Haihe Biology is a typical representative of ODM services. The platform has a manufacturing service center of over 3,000 square meters, equipped with 100,000-level, 10,000-level and local 100,000-level clean workshops, providing a complete solution from product research and development, sample production, to design customization of intelligent production lines, and post-market contract manufacturing.
The core value of ODM services lies in technology integration and resource optimization. Manufacturers incorporate their technological accumulation, production experience, and supply chain resources into the product development of their clients, thereby shortening the development cycle and reducing development risks. Professional teams possess outstanding design conversion capabilities, enabling them to transform clinical needs into manufacturable product designs.
Intellectual property protection is a key aspect of ODM services. Manufacturers need to establish a sound intellectual property management system, clarify the ownership of all customer designs, and protect customers' innovative achievements. At the same time, the manufacturer's own patents and technical secrets also need to be protected to prevent the leakage of core competitiveness during cooperation.
The CDMO Model: A New Trend in Contract Development and Manufacturing Organizations
The CDMO (Contract Development and Manufacturing Organization) model is receiving increasing attention in the medical device field. Compared with the traditional CMO (Contract Manufacturing Organization), CDMO gets involved in the product development stage earlier and provides comprehensive services from concept to commercialization.
The Haihe Bio CDMO platform offers services including contract design, development and production, technology transfer, inspection and testing, clinical trials, pre-market research, scientific research outsourcing, supply chain establishment, regulatory support, and high-quality contract manufacturing. By seamlessly integrating all business segments, the platform can flexibly adjust service contents based on customer requirements to ensure precise matching with customer needs.
The CDMO model is particularly suitable for the development of innovative medical devices. These products are often technologically complex, subject to high regulatory requirements, and carry significant development risks. CDMO providers, with their rich industry experience and professional teams, can help clients overcome technical challenges and regulatory hurdles, thereby increasing the success rate of product launches.
Personalized customization and special project services
For some special needs, manufacturers offer in-depth personalized customization services. INVAMED's Customization and Special Projects Department is dedicated to handling highly specialized medical challenges and critical mission requirements. From medical equipment for deep space exploration to ultra-specific R&D plans, to one-of-a-kind medical device prototypes, this service breaks through the boundaries of traditional medical devices.
Interdisciplinary collaboration is a hallmark of special project services. Manufacturers integrate expertise from physics, engineering, and life sciences to meet unprecedented demands for medical devices. Technologies such as rapid prototyping, 3D printing, and novel material testing are employed to validate concepts in the earliest stages of custom product development.
Extreme environment applications represent another specialized field. Medical devices designed for space missions must be capable of withstanding cosmic radiation, vacuum conditions, and extreme temperature fluctuations; tools and equipment designed for high-altitude, underwater, polar, or desert conditions need to meet medical demands in harsh environments.
Technical support and training services
A well-developed after-sales service is an important component of customized services. Product usage training helps medical staff operate and maintain equipment correctly, reducing the risk of operational errors and equipment damage. Technical support and maintenance services ensure that any issues that arise during the use of the products can be resolved promptly, minimizing the risk of surgical interruptions.
Some manufacturers also offer surgical plan consultation and optimization services. Based on a deep understanding of the products and clinical applications, they assist medical institutions in formulating the best surgical instrument configuration plans to enhance surgical efficiency and safety. Regular follow-ups and user feedback collection provide a basis for product improvement and new product development.
Global layout and localized services
With the globalization of the medical device market, the global layout capability of manufacturers has become particularly important. Mindray Medical's internationalization strategy is worth learning from. It has planned local production in 14 overseas countries, and 11 of them have already started production. Through the acquisition of the world's top in vitro diagnostic raw material suppliers and European diagnostic companies, Mindray has not only strengthened its independent control over the upstream of the industrial chain but also quickly built a localized supply chain capability to serve the European market.
Localized production and services can better meet the demands of regional markets, shorten the supply cycle, and reduce logistics costs. At the same time, local teams have a better understanding of local regulations, cultures, and clinical practices, and can provide more appropriate technical support and customer service.
Digital Transformation and Intelligent Services
Digital technology is transforming the model of customized services. Online configuration tools enable customers to select product specifications, materials and functions through web interfaces, and view product effects and prices in real time. Virtual reality and augmented reality technologies help customers experience the product usage effects before purchase, reducing decision-making uncertainty.
The Internet of Things (IoT) technology enables real-time collection and analysis of product usage data. By embedding sensors and connecting to cloud platforms, manufacturers can monitor product performance, usage frequency, and maintenance needs, providing predictive maintenance and optimization suggestions. Big data analysis helps manufacturers understand product usage patterns and changes in customer needs, guiding new product development and service improvements.
Future Development Trends and Challenges
Modular design will become an important direction for customized services. By combining standardized modules, products that meet different needs can be quickly configured, balancing personalization and cost efficiency. The platform strategy enables manufacturers to develop multiple products based on the same technical platform, sharing R&D achievements and production resources.
Sustainable development poses new challenges for customized services. The selection of eco-friendly materials, energy conservation and emission reduction in the production process, and the recyclable design of products all need to be incorporated into product development and service provision. Manufacturers need to assist customers in achieving a balance between economic and environmental benefits.
Artificial intelligence and machine learning technologies will further enhance the level of customized services. Intelligent recommendation systems suggest the most suitable product configurations based on customers' historical orders and industry trends; intelligent design tools automatically generate product design plans that meet technical requirements; and intelligent supply chain management systems optimize inventory and logistics to improve response speed.
Overall, the customization services of laparoscopic trocar manufacturers are shifting from simple production based on drawings to comprehensive solutions. Technical capabilities, service quality, and response speed have become the key competitive factors. Manufacturers need to build a flexible production system, a professional technical team, and a complete service network, while embracing digital and intelligent technologies, in order to establish a sustainable competitive advantage in the field of customization services.